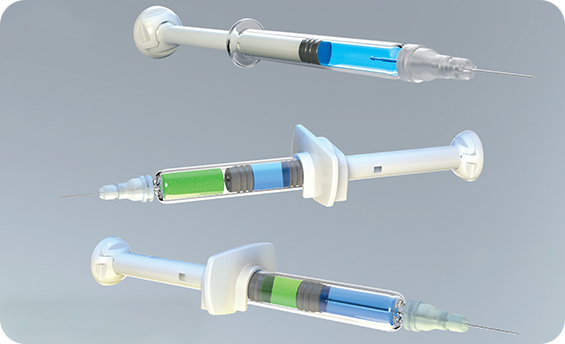
Syringe Scale Line Verification Vision Measurement Systems
Ensuring Precision and Compliance
In the pharmaceutical industry, syringe production, particularly for injectable use, demands rigorous quality control. Syringe scale line verification vision measurement systems ensure syringes meet precise specifications and comply with safety and regulatory standards. Leveraging advanced machine vision, AI-based measurement tools, and high-precision optics, guarantees accurate scale line verification, minimizing defects and production errors.
Advanced Image Processing for Accurate Line Detection
Vision systems employ several advanced image processing techniques tailored for scale line verification:
Edge Detection

Line Segmentation

Pattern Recognition

Recognize various scale line patterns, such as numbered increments, graduated lines, and fine markings, even if the patterns vary across syringe types or manufacturers.

Precision Measurement
Using High-Resolution Imaging Technology
The Syringe Scale Line Verification system utilizes industrial-grade cameras with sub-micron resolution to capture precise images of syringe scale lines. Image processing algorithms analyze the position, width, and spacing of the scale lines, using edge detection and contour analysis to compare them against standards for accurate verification.
Seamless Integration with Production Lines
Optimizing Workflow with Monitoring
Our vision measurement systems are engineered for seamless integration into existing production lines. Whether your production environment is high-volume or low-volume, the system can be easily incorporated into your workflow, interfacing with other machines and quality control systems.

Regulatory Compliance and Industry Standards
Ensuring Precision and Compliance
Syringe manufacturing is subject to strict regulatory standards to ensure patient safety. Our Syringe Scale Line Verification Vision Measurement Systems are compliant with industry regulations ISO 13485, FDA 21 CFR Part 820, and CE marking requirements. The system's ability to generate traceable, auditable records ensures that your production processes meet the required compliance standards for medical devices.
 AucoVision Automated Bottle Ne
AucoVision Automated Bottle Ne
 Automated Cap Color Consistenc
Automated Cap Color Consistenc
 Auto Medicine Carton Box Damag
Auto Medicine Carton Box Damag
 Syringe Scale Line Verificatio
Syringe Scale Line Verificatio